This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
Protein domains of COL1A1
PFAM
There were 15 PFAM matches with the FASTA protein sequence. The following is a list of the three domains found:
SMART
Three different types of domains were found using SMART analysis:
PROSITE
The von Willebrand factor type C repeat domain was found twice within this protein using PROSITE. According to this analysis, this is a domain that is 70 amino acids long with 10 cysteines. This domain is commonly found in other proteins known to form large complexes, further supporting the hypothesis that this domain may be involved in complex formation. [2,3]
There were 15 PFAM matches with the FASTA protein sequence. The following is a list of the three domains found:
- von Willebrand factor type C domain (VWC)
- Collagen triple helix repeat (found 12 times)
- Fibrillar collagen C-terminal domain (COLFI)
SMART
Three different types of domains were found using SMART analysis:
- VWC
- COLFI
- Low complexity (LC)
PROSITE
The von Willebrand factor type C repeat domain was found twice within this protein using PROSITE. According to this analysis, this is a domain that is 70 amino acids long with 10 cysteines. This domain is commonly found in other proteins known to form large complexes, further supporting the hypothesis that this domain may be involved in complex formation. [2,3]
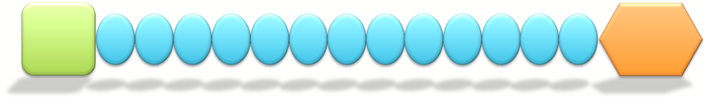
Figure 1. Hypothetical layout of domains found in COL1A1. Green domain on the left represents the VWC domain, while the orange domain represents the COLFI domain. The remaining blue oval domains in between represent each collagen triple helix repeat domain.
References
1. Bork P; , FEBS Lett
1992;307:49-54.: The modular architecture of vertebrate collagens.
2. Bork P; , FEBS Lett 1993;327:125-130.: The modular architecture of a new family of growth regulators related to connective tissue growth factor.
3. Hunt, L., and W. Barker. "Von Willebrand Factor Shares a Distinctive Cysteine-rich Domain with Thrombospondin and Procollagen." Biochemical and Biophysical Research Communications 144.2 (1987): 876-82.
4. O'Leary, J.M. et al. "Solution Structure and Dynamics of a Prototypical Chordin-like Cysteine-rich Repeat (von Willebrand Factor Type C Module) from Collagen IIA." J Bio Chem. Web. 10 Feb. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/15466413>.
5. Kuivaniemi H, Tromp G, Prockop DJ Mutations in fibrillar collagens (types I, II, III, and XI), fibril-associated collagen (type IX), and network-forming collagen (type X) cause a spectrum of diseases of bone, cartilage, and blood vessels. Hum Mutat. 1997; 9: 300-15
6. Mayne R, Brewton RG; , Curr Opin Cell Biol 1993;5:883-890.: New members of the collagen superfamily.
2. Bork P; , FEBS Lett 1993;327:125-130.: The modular architecture of a new family of growth regulators related to connective tissue growth factor.
3. Hunt, L., and W. Barker. "Von Willebrand Factor Shares a Distinctive Cysteine-rich Domain with Thrombospondin and Procollagen." Biochemical and Biophysical Research Communications 144.2 (1987): 876-82.
4. O'Leary, J.M. et al. "Solution Structure and Dynamics of a Prototypical Chordin-like Cysteine-rich Repeat (von Willebrand Factor Type C Module) from Collagen IIA." J Bio Chem. Web. 10 Feb. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/15466413>.
5. Kuivaniemi H, Tromp G, Prockop DJ Mutations in fibrillar collagens (types I, II, III, and XI), fibril-associated collagen (type IX), and network-forming collagen (type X) cause a spectrum of diseases of bone, cartilage, and blood vessels. Hum Mutat. 1997; 9: 300-15
6. Mayne R, Brewton RG; , Curr Opin Cell Biol 1993;5:883-890.: New members of the collagen superfamily.